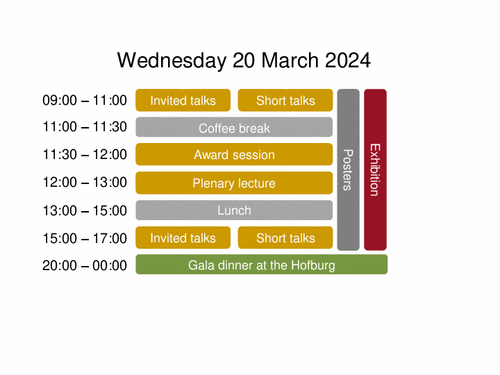
Invited talks: Innovations in oral drug delivery
| 09:00-09:40 | Amorphous solid dispersion release mechanisms Lynne Taylor, Purdue University, USA |
| 09:40-10:20 | Oral delivery of systemic monoclonal antibodies, peptides and small molecules using gastric auto- injectors Alex Abramson, Georgia Institute of Technology, USA |
| 10:20-11:00 | Oral delivery and intestinal targeting of therapeutic nucleic acids Caitriona O‘Driscoll, University of Cork, Ireland |
Invited talks: Parenteral controlled drug delivery
| 09:00-09:40 | Noninvasive in vitro and in vivo monitoring of parenteral controlled release DDS Karsten Maeder, University of Halle, Germany |
| 09:40-10:20 | Changing the rules in the tumor microenvironment. Developing and delivering nanoparticle catalysts to growing tumors Jesús Santamaría Ramiro, University of Zaragoza, Spain |
| 10:20-11:00 | Injectable hydrogels for drug delivery Tina Vermonden, University of Utrecht, The Netherlands |
Short talks: Paediatric drug delivery
| 09:00-09:20 | Hazzle-Free Pediatric Pain Treatment via Personalized Inkjet-Printed Temporary Tattoos Martin Spörk, Graz University of Technology, Graz, Austria |
| 09:20-09:40 | Towards a standard vehicle toolbox: development and verification of standard vehicles for compatibility studies with oral paediatric drug products Carolin Eckert, University of Greifswald, Germany |
| 09:40-10:00 | Development of an oral solution of vinorelbine to protect the oral mucosa during paediatric administration François Xavier Legrand, Université Paris Saclay, France |
Short talks: Excipients and raw materials
| 10:00-10:20 | Suitability Study on Microcrystalline Cellulose for Tablets Containing Pressure-Sensitive Substances Tomohisa Fujisawa, Asahi Kasei Corporation, Noboeoka, Japan |
| 10:20-10:40 | A nitrosamine calculation tool to support risk mitigation Sara Fathollahi, DFE Pharma, Germany |
| 10:40-11:00 | Croscarmellose-sodium as pelletization aid - the role of multivalent cations Finn Siebel, Heinrich Heine University, Düsseldorf, Germany |
Short talks: Physical pharmacy and preformulation
| 09:00-09:20 | The enigmatic world of hydrochlorothiazide: polyamorphs and their phase interconversions Inês Martins, University of Copenhagen, Denmark |
| 09:20-09:40 | Development of an oral liquid formulation of ONC201 to limit its degradation and increase its aqueous solubility Maxime Annereau, Gustave Roussy Cancer Campus, Villejuif, France |
| 09:40-10:00 | Nucleation of celecoxib glass: Determination of nucleation temperature and impact of nuclei on dissolution/physical stability performances Kohsaku Kawakami, National Institute for Material Science, Tsukuba, Japan |
Short talks: Quality control and PAT
| 10:00-10:20 | Real-time particle size measurement during fluidized bed processes using a convolutional neural network-based endoscopic imaging system Orsolya Péterfi, Budapest University of Technology and Economics, Budapest, Hungary |
| 10:20-10:40 | Digital Real-time Release Testing in Continuous Tablet Production Lines Selma Celikovic, Research Center Pharmaceutical Engineering GmbH, Graz, Austria |
| 10:40-11:00 | Influences of process parameters on the wet film thickness in continuous manufacturing of oral films Leonie-Lara Dorothea Uth, Heinrich Heine University, Düsseldorf, Germany |
APGI award session
Plenary lecture
| 12:00-13:00 | Observations which might inspire drug delivery scientists for guiding bio-therapeutics over biological barriers Stefaan de Smedt, University of Ghent, Belgium |
Invited talks: Continuous manufacturing and PAT
| 15:00-15:40 | Continuous Manufacturing of Oral Solid Dosage Forms: what’s next ? Bart Nitert, Janssen Pharmaceuticals, Beerse, Belgium |
| 15:40-16:20 | Innovative continuous pharmaceutical technologies supported by AI and machine vision Zsombor Kristóf Nagy, University of Budapest, Hungary |
| 16:20-17:00 | CPV 4.0: An industrial app to enhance the CPV (Continuous Process Verification) Marc Ramoneda, Appliant – Apps GMP Compliant S.L., Spain |
Invited talks: Cell and gene therapies
| 15:00-15:40 | ATMPs on the move: overview and regulatory perspective Ralf Sanzenbacher, Paul Ehrlich Institute, Germany |
| 15:40-16:20 | Ex vivo CAR-T cell concept & regulatory considerations Sergio Fracchia, Novartis, Switzerland |
| 16:20-16:40 | In vitro and in vivo delivery kinetics of ionizable lipid nanoparticles encapsulating TNF-α siRNA and their efficacy on inflammatory models Qinglin Wang, University of Paris-Saclay, France |
| 16:40-17:00 | Anti-miRNA103/107 lipid nanoparticles for the treatment of brain ischemic damage Virginia Campani, University of Naples Federico II, Italy |
Short talks: Bioavailability, absorption enhancement and in-vitro/in-vivo correlations
| 15:00-15:20 | Dissolution vs Solubility Mismatch in Intestinal Buffer Jozef Al-Gousous, Johannes Gutenberg University Mainz, Germany |
| 15:20-15:40 | Novel simulated interstitial fluid media for predictive subcutaneous in vitro assays Iria Torres Terán, University of Greifswald/Sanofi Frankfurt, Germany |
| 15:40-16:00 | In vitro Dissolution/Permeation tools to forecast ASDs bioavailability: comparison and mechanistic insights Patricia D. Nunes, Hovione Farmaciência S.A., Lisbon, Portugal |
Short talks: Topical and transdermal delivery
| 16:00-16:20 | Design of a novel PVA/PVP/PEGdiacid-based hydrogel-forming microneedle array patches for enhanced and sustained transdermal delivery of enfuvirtide Huanhuan Li, Queen´s University Belfast, United Kingdom |
| 16:20-16:40 | Thermotropic liquid crystals: an intelligent strategy for skin drug delivery Mariia Nesterkina, Helmholtz Institute for Pharmaceutical Research Saarland, Saarbrücken, Germany |
| 16:40-17:00 | Novel method for testing antimicrobial effectiveness of local drug-delivery systems Kelli Randmäe, University of Tartu, Estonia |
Short talks: Nanoparticles and Vesicles II
| 15:00-15:20 | Biomimetic vesicles for glioblastoma treatment Giorgia Ailuno, University of Genoa, Italy |
| 15:20-15:40 | Harnessing the potential of tissue-derived extracellular vesicle therapy for precision regenerative medicine Peng Lou, West China Hospital of Sichuan University, China |
| 15:40-16:00 | A novel multidrug nanoparticle formulation assessment in a cardiac ischemia reperfusion model Natalie Lan Linh Tran, University of Paris Saclay and University of Namur, France |
| 16:00-16:20 | Reproducible Synthesis of Biocompatible Albumin Nanoparticles Designed for Intra-Articular Delivery of Celecoxib to treat Osteoarthritis Rumi Khandelia, University College Dublin, Ireland |
| 16:20-16:40 | Microfluidic development of biomimetic hybrid nanoparticles for homologous-targeting and enhanced therapy of cancer Ilaria Arduino, University of Bari, Italy |
| 16:40-17:00 | Antioxidant-Based Nanoparticles Protect against Intracellular Reactive Oxygen Species in Neurological Disorders Thelma Akanchise, University of Paris Saclay, France |

